|
In addition to eIF4F and eIF2, multiple other initiation factors facilitate the formation of the 48S initiation complex. The 40S complex subsequently scans down the mRNA until encountering the first AUG and the 48S initiation complex forms. 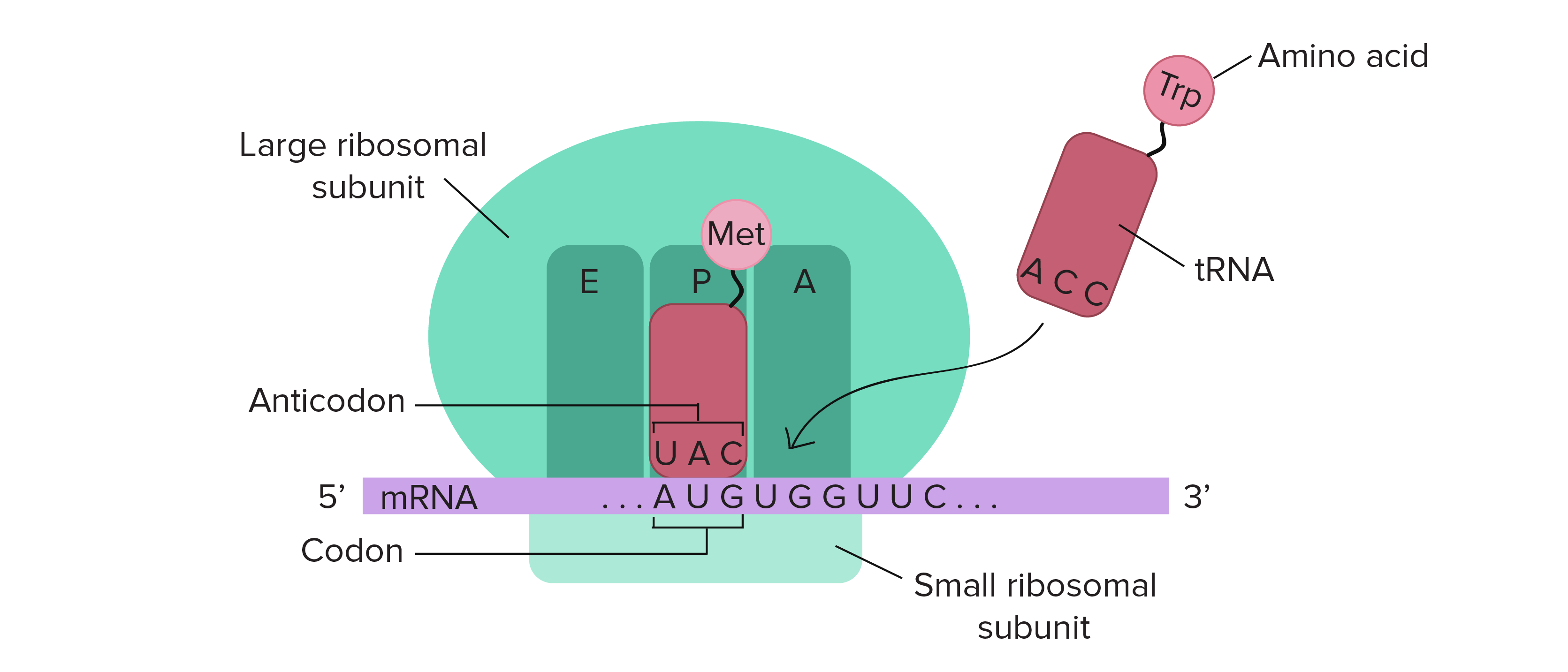
The eIF4F complex then recruits the 43S pre-initiation complex comprised of 40S subunits together with a ternary complex formed of the initiator tRNA (Met-tRNA), eIF2, and GTP to the 5’ end of an mRNA. A protein complex known as eIF4F recognizes the cap structure. In eukaryotes, the AUG used to initiate protein synthesis is usually the first AUG downstream of the cap structure, found at the 5’ end of the mRNA. 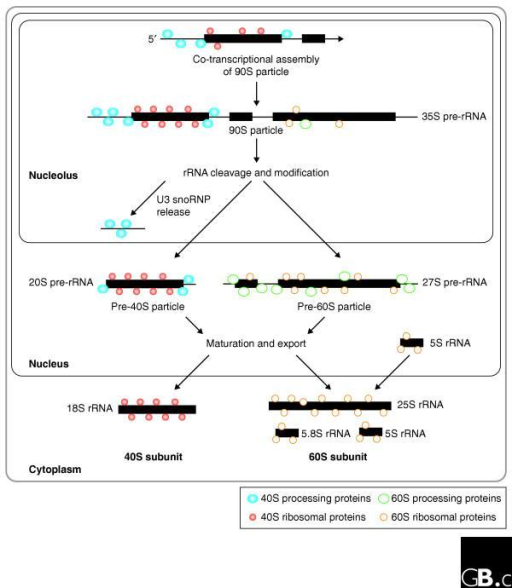
The AUG codon specifies the amino acid methionine and virtually all proteins specified by the genetic code begin with methionine. This process involves identifying appropriate codon to initiate translation. Translation begins with the assembly of an 80S initiation complex on mRNA. Finally, termination occurs when the ribosome encounters one of three termination codons, and the completed protein gets released from the ribosome. Upon identification of the start site for translation, elongation ensues as the ribosome moves along the mRNA “reading” groups of three nucleotides that specify each amino acid added to the growing polypeptide chain. This process has significant differences between eukaryotes (described here) and prokaryotes. Initiation consists of identifying the exact site in the sequence of nucleotides in an mRNA to begin translation. īriefly, translation can be broken down into three phases initiation, elongation, and termination. In contrast, ribosomes translating mRNAs encoding proteins destined for secretion from the cell or resident proteins of the endoplasmic reticulum, Golgi apparatus, lysosome, or plasma membrane get localized to the endoplasmic reticulum membrane. Ribosomes translating most cellular mRNAs do so as free ribosomes in the cytoplasm. While most of the steps involved in maturing ribosomal subunits occur within the nucleolus before the subunits exiting through nuclear pores, final steps in subunit maturation occur in the cytoplasm. Ribosome assembly is a complex process involving hundreds of accessory factors that transiently associate with ribosomal subunits during their maturation. This assembly process defines a compartment of nucleus referred to as the nucleolus. Ribosomal proteins then return to the nucleus where they assemble hierarchically on rRNAs being transcribed by RNA polymerase I. Transfer RNAs and the mRNAs encoding ribosomal proteins exit the nucleus and the latter get translated in the cytoplasm. The synthesis of components of the translational machinery begins with the transcription of mRNAs, tRNAs, and rRNAs in the nucleus by RNA polymerases II, III, and I, respectively. The eukaryotic cell is compartmentalized, with different cellular compartments defined by biological membranes. As our understanding of the mechanisms of protein synthesis continues to grow, there will likely be countless additional applications for this knowledge in medicine, research, and industry. Science has modified many of these compounds that target the machinery for protein synthesis in pathogenic microbes for use in the clinic as antibiotics. 
These differences have been exploited in nature as organisms produce compounds targeting the protein synthetic machinery of competitors as they vie for limited resources. Despite having certain underlying similarities in their mechanism, protein synthesis in the three major lines of descent (bacteria, archaea, and eukaryotes) has diverged to the point that substantive mechanistic differences have arisen. As one of the foundational concepts in biology, protein synthesis is sufficiently complex that many believe it evolved once, giving the protein synthetic machinery in all organisms on the planet a common ancestry. Understanding protein synthesis is paramount in studying various medical fields, from the molecular basis of genetic diseases through antibiotic development to expressing recombinant proteins as drugs or clinical laboratory reagents. In the last few decades, advances in laboratory techniques for the study of these microscopic sciences have led us to a greater understanding of the central dogma of molecular biology – that DNA transcribes RNA which then gets translated into protein. 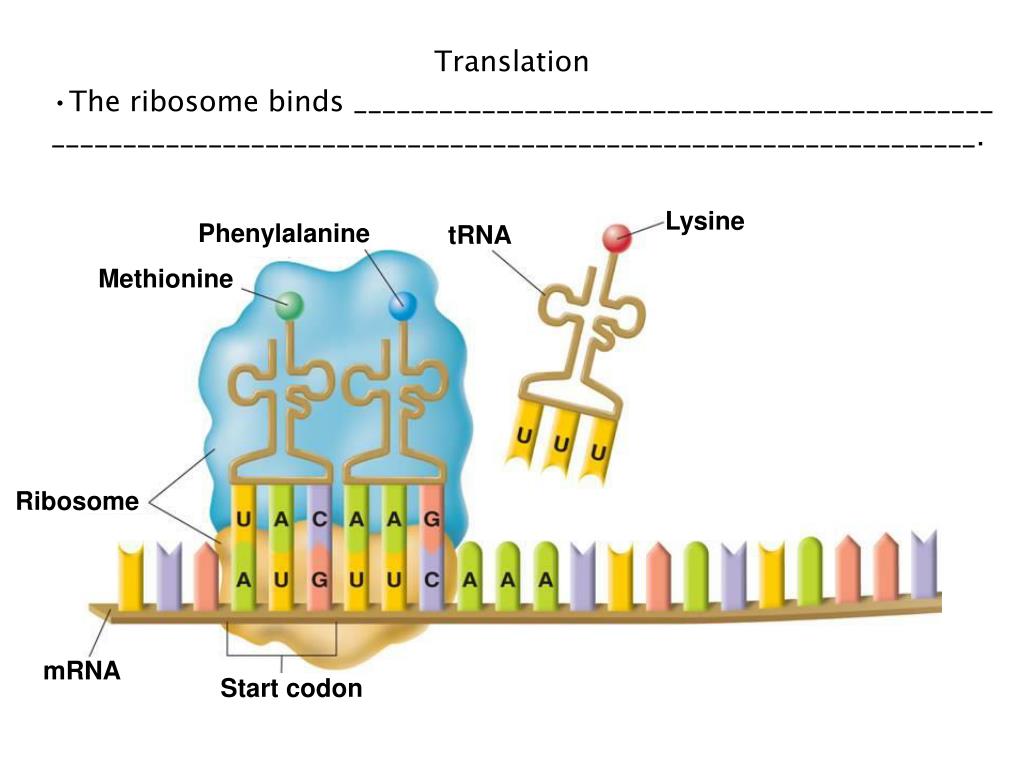
Our understanding of each of the biological sciences becomes heightened by the study of biochemistry and molecular biology.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
